Amine gas treatment also known as Gas sweetening, Amine scrubbing, or acid gas treatment is the process that removes H2S/CO2 and mercaptan compounds from the acid gas streams by absorption into aqueous solutions called Alkanolamines or amines. Amine treating is the most widely used in oil refineries, petrochemicals, and natural gas processing plants for removing acid gases from the process gas streams. To avoid foaming in Amine System please view the Blog “Control of Foaming in Amine System“.
Acid Gas Treatment in the oil and gas serves some major purposes as follows;
- H2S removal from the recycle gas streams of Hydrotreating and hydrocracking units to maintain the purity of the reactor feed gas.
- Treatment of the acid gases produced from the stripping sections of hydroprocessing units.
- Treatment of refinery fuel gas to prevent corrosion problems and increase the heating value of the gas.
- Treatment of liquefied petroleum gas before Merox process.
- Removal of CO2 from synthesis gas at hydrogen production plants.
- Removal of H2S/CO2 from the natural gas streams.
Amine Treating Process Description
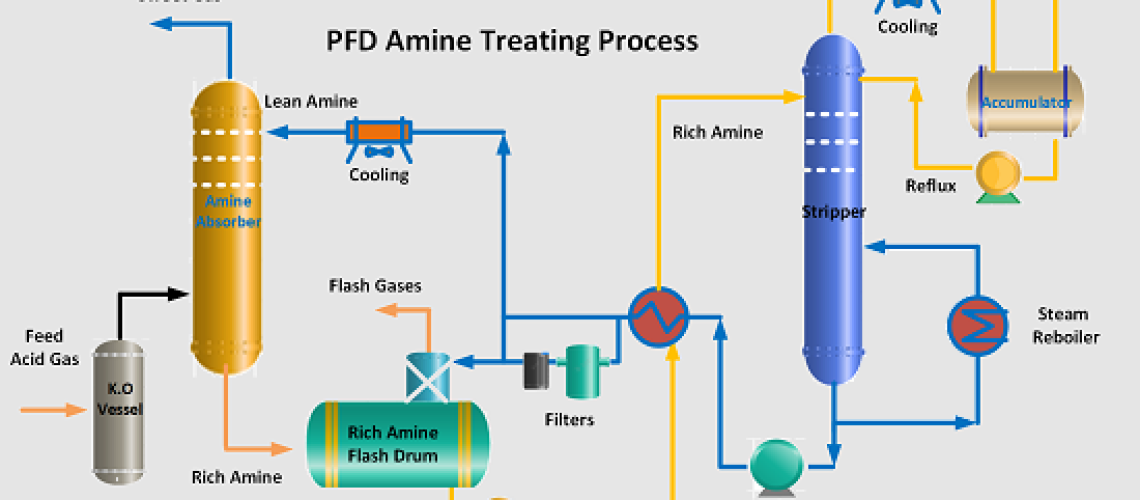
A typical amine process is shown in the Figure below. The acid feed gas first enters the knock-out vessel to remove any liquid hydrocarbons. The gas then enters the bottom of the Amine Scrubber or Amine Absorption tower which has either trays or a packed bed. Tray towers are applied for high flow rates while packed columns are for reduced flow rates.
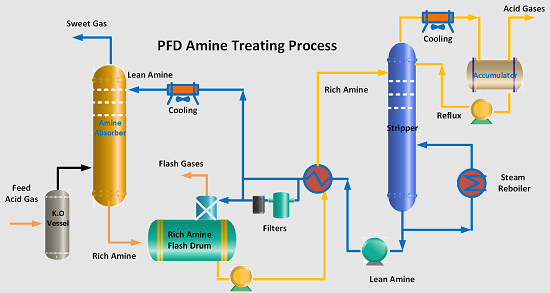
The sour gas moves from the bottom to the top of the absorption tower. The lean or regenerated amine enters at the top of this tower and two streams are contacted counter currently. Acid components of the sour gas (CO2/H2S) are absorbed with the chemical reaction into the amine phase. The bottom exit amine solution, loaded with CO2 and H2S, is called a rich amine.
This rich amine solution is first flashed in a flash drum to separate any hydrocarbons from the rich amine stream. The separated hydrocarbon is removed from the flash drum and recycled as slop oil. The flashed gases are normally used as fuel. A small packing tower is installed on top of the flash drum to recover H2S from the flashed hydrocarbon gas by counteracting with a slipstream of lean amine.
The rich amine from the flash drum is pumped and preheated with the stripper lean-rich heat exchanger. The heated rich amine enters the top of the stripper to strip out acid gases (CO2 and H2S) from the solution and exits at the top of the tower. Steam-heated reboilers are applied at the bottom of the stripper to regenerate the rich amine. A small quantity of live steam is also injected into the stripper to achieve the water balance in the system.
The lean regenerated from the stripper, after partial cooling in the lean-to-rich solution heat exchanger, is pumped to the amine absorber through an air-cooled exchanger. A slipstream of 10 ~15% is passed through mechanical filters to remove solid particles and carbon filters to remove chemical impurities from the lean amine solution.
Acid gases stripping column top are cooled to condense a major portion of the water vapor. This condensate is continually fed back to the system to prevent the amine solution from becoming progressively more concentrated. Generally, all of this lean solution is fed back to the top of the stripping column at a point above the rich-solution feed and serves to absorb and return amine vapors carried by the acid gas stream.
The regenerated amine (lean amine) is recycled back to the top of the absorption tower. The stripped overhead gas from the regenerator is concentrated in H2S and CO2. This H2S-rich stripped gas stream is then usually sent to a Claus process to convert it into elemental Sulphur.
For further information, discussion and queries please comment in the box below or contact us at admin@ or follow us on Facebook & LinkedIn.
Certified Functional Safety Professional (FSP, TÜV SÜD Germany), Certified HAZOP & PHA Facilitator, LOPA Practitioner, QRA/FERA and Specialist in SIL Verification & Functional Safety Lifecycle, with 18 years of professional experience in Plant Process Safety across Petroleum Refining and Fertilizer Complexes.
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain