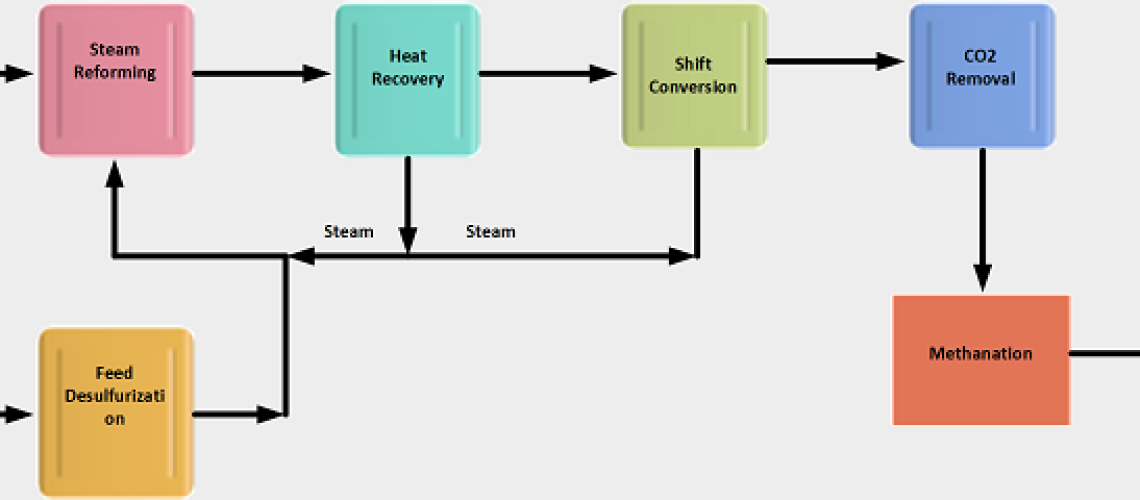
Steam reforming is a method for producing hydrogen gas by the reaction of hydrocarbon in the presence of steam and catalyst. Natural gas, refinery gas, liquefied petroleum gas (LPG), and low-boiling naphtha are some hydrocarbons with low sulfur contents frequently used as feedstocks for steam reforming. Hydrogen produced by steam reforming is termed ‘grey hydrogen’ when the byproduct carbon dioxide gas is released into the atmosphere and ‘blue hydrogen’ when the carbon dioxide is mostly captured, stored, and utilized.
Hydrogen is the most valuable energy source for future oil refineries to produce the best quality petroleum products. Hydrogen is the backbone of petroleum oil refinery and is necessary to hydrotreat the finished petroleum products or the feeds of other refinery units and to hydrocrack the heavier gas oils into distillates. The hydrogen produced during steam reforming can be used as a feed for hydrocarbon hydroprocessing (hydrotreating, and hydrocracking ) fuel for fuel cells, or as a byproduct in manufacturing chemicals like methanol, ammonia, and cryogenics.
1. Desulfurization of Feedstock
The most common feedstock for producing hydrogen is natural gas because it satisfies all reformer feedstock specifications, is more readily available, and has higher calorific values than other feedstocks. If heavy hydrocarbons are used then the pre-reforming of feed is performed to break down higher hydrocarbons such as propane, butane, or naphtha into methane (CH4), which allows for more efficient reforming downstream. The methane feedstock must be hydrotreated to remove sulfur compounds and other impurities. This process turns organic Sulphur into hydrogen, in the presence of CoMo catalyst at a 350~400 °C temperature, and is called Hydrodesulfurization.
CH3SH + H2 CH4 + H2S
H2S produced is then reacted with zinc oxide (ZnO) at a temperature of about 370 °C (700 °F), ensuring complete hydrogenation and maximizing the use of the zinc oxide.

2. Steam Reforming of Feedstock
Low-boiling hydrocarbons, like methane, react with steam in steam reforming to produce hydrogen. Typically, the reaction takes place over a nickel catalyst packed into the tubes of a reforming furnace at about 700~1000 °C. In steam methane reforming, methane reacts with steam under 25~35 bar pressure in the presence of a steam methane reforming catalyst to produce hydrogen, carbon monoxide, and a relatively small amount of carbon dioxide. The steam methane reforming process for hydrogen production is endothermic and a significant amount of heat is supplied to the process for the reaction to proceed. The steam methane reforming reaction equation is shown below;
CH4 + H2O → 3H2 + CO
CO+H2O→CO2+H2
The equation for the chemical balance of higher-boiling-point hydrocarbons expressed in a more general form is CnHm + nH2O → (n + m/2) H2 + nCO
The hydrocarbon feedstock also undergoes a series of cracking reactions at high temperatures. In addition, carbon further reacts with steam. In natural gas steam reforming, high excess of steam is required, expressed by the (molar) steam-to-carbon (S/C) ratio. Typical steam to carbon ratio values lie within the range 2.5:1 – 3:1
CH4 → 2H2 + C
C + H2O → CO + H2
The steam reforming reaction is conducted in multitubular catalyst-packed bed reactors, a subtype of the plug flow reactor category. These reactors consist of an array of long and narrow tubes which are situated within the combustion chamber of a large industrial furnace, providing the necessary energy to keep the reactor at a constant temperature during operation. Steam reforming of natural gas is 65–75% efficient.
Inside the tubes, a mixture of steam and methane is put into contact with a nickel catalyst. Catalysts with a high surface-area-to-volume ratio are preferred because of diffusion limitations due to high operating temperatures. Examples of catalyst shapes used are spoked wheels, gear wheels, and rings with holes. Additionally, these shapes have a low-pressure drop which is advantageous for this application.
Hydrocarbons are converted to hydrogen and carbon monoxide on the catalyst simultaneously as carbon is produced. During steam reforming of natural gas or similar feedstock, the carbon can be quickly removed by reaction with steam. When higher-boiling feedstocks are used, the carbon is not removed quickly enough and accumulates, requiring either catalyst replacement or regeneration. Further, the heat of the reaction is recovered by producing steam in the reboilers which is then used in the reforming and shift reactions as a feed.
3. Water Gas Shift Reactions
After reforming, the synthesis gas contains a sufficient amount of CO (12~15%) & CO2 (5~10%) along with hydrogen. The convenient way to remove carbon monoxide is to oxidize it to carbon dioxide, which can then be removed by an absorption process. The carbon monoxide is oxidized to carbon dioxide by steam along with the production of more Hydrogen. The reaction is known as the water gas shift reaction and produces more hydrogen. Further, shift reactions are performed in two steps called low-temperature and high-temperature shift reactions. The water gas shift reaction is shown below;
CO + H2O → CO2 + H2
Steam methane reforming plants use copper/zinc low-temperature shift catalysts and iron/chrome high-temperature shift catalysts as their primary shift catalysts. Magnetite (Fe3O4) is the main component of high-temperature shift catalysts, which operate in the range of 315°C–430°C (600°F–800°F), with three-valent chromium oxide (Cr2O3) added as a stabilizer. Low-temperature shift catalysts function between 205°C and 230°C (400°F and 450°F). The reaction equilibrium is easier to control, and less carbon monoxide is produced at the lower temperature.
4. CO2 Removal
After the conversion of all CO into CO2, the Carbon Dioxide (CO2) is removed either by “pressure-swing adsorption,” or by absorption in Amine. The carbon dioxide and other impurities are removed from the gas stream, leaving essentially pure hydrogen.
5. Methanation Reactions
The synthesis gas that contains the remaining CO (Carbon Monoxide) & CO2 (Carbon dioxide) are converted to methane in the presence of steam and catalyst. The methanation process uses a catalyst which is nickel on an alumina carrier. The catalyst has a long life because it operates in ideal conditions and is not poisoned. The iron oxide catalyst reacts with the gas at a temperature range of 315°C to 370°C (600°F to 700°F); The reaction is highly exothermic, any change in the feed composition and rise in mainly CO2 will suddenly increase the temperature.
CO + 3H2O → CH4 + H2O
CO2 + 4H2 → CH4 + 2H2O
Top References
- Refining Process Handbook by Surinder Parkash.
- Handbook of Petroleum Refining by James G. Speight.
- Article: Current Status of Hydrogen Production Techniques by Steam Reforming of Ethanol: A Review.
- Fundamentals of Petroleum Refinery by M.A Fahim.
- Article: Thermodynamic analysis of hydrogen production by steam reforming.
- Fundamentals of Petroleum and Petrochemical Engineering by Uttam Ray Chaudhuri
For further information, discussion and queries please comment in the box below or contact us at admin@ or follow us on Facebook & LinkedIn.
Certified Functional Safety Professional (FSP, TÜV SÜD Germany), Certified HAZOP & PHA Facilitator, LOPA Practitioner, QRA/FERA and Specialist in SIL Verification & Functional Safety Lifecycle, with 18 years of professional experience in Plant Process Safety across Petroleum Refining and Fertilizer Complexes.
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain