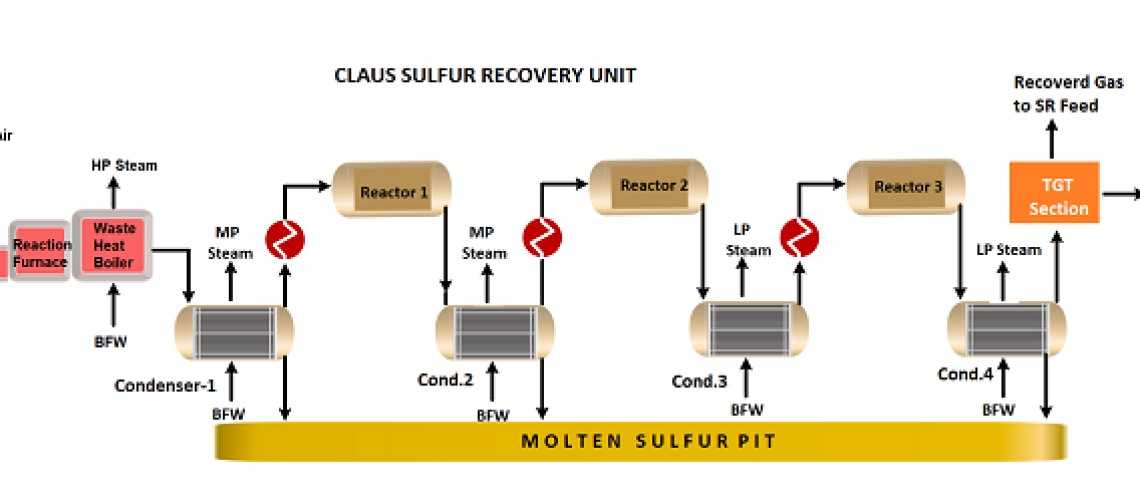
The Sulfur Recovery Unit (SRU) in a Petroleum Refinery is designed to remove H2S from sour gases originating in the various refinery processes and convert them to industrial-grade molten sulfur. The process used in the sulfur recovery units in the petroleum oil refineries is known as the “Claus Process”, which removes 95-99.9 % of hydrogen sulfide present in the gas streams. The modified Claus Sulfur recovery process burns the H2S-rich sour gas with oxygen, cools the burnt gas, and recovers molten sulfur from the burnt gas.
Sulfur recovery units in oil refineries are applied to remove H2S from the refinery processes to meet environmental regulations related to SOX and H2S emissions and reduce the environmental pollution related to petroleum products. Moreover, owing to stringent environmental regulations and the implementation of Euro fuel quality standards for diesel, gasoline, and other petroleum products have increased the requirement for Sulfur Recovery Units (SRU).
Sulfur compounds present in crude oil are transferred to all products fractionated in a crude distillation unit. Sour crudes contain sulfur more than 0.5% of compared to sweet crudes. In the downstream units, Sulfur compounds are converted to H2S and produce sour gases which are then absorbed in the Amine solution. The following are sources of H2S in the petroleum oil refinery;
- Refinery off gases, LPG separated from Crude Distillation unit and Gas Concentration Unit, recycle gases of hydrotreating units and Hydrocracking units. These gas streams are treated with an amine to remove H2S in an Amine Gas Treating Unit. Regeneration of rich amine produces feed gas for the sulfur recovery unit.
- Sour water produced in Hydrotreating and hydrocracking units is strapped to produce H2S and ammonia-rich gases which are also fed to the SRU (Sulfur Recovery Unit).
Sulfur Recovery Unit Process Description
The Claus Sulfur Recovery Unit in an oil refinery is divided into three steps the Sulfur Recovery Section, the Tail Gas Treating (TGT) Section, and Incineration section. Claus Sulfur recovery unit process works to convert hydrogen sulfide (H2S) gas from the refinery sour gases to elemental sulfur. Claus sulfur recovery unit process flow diagram is shown below;
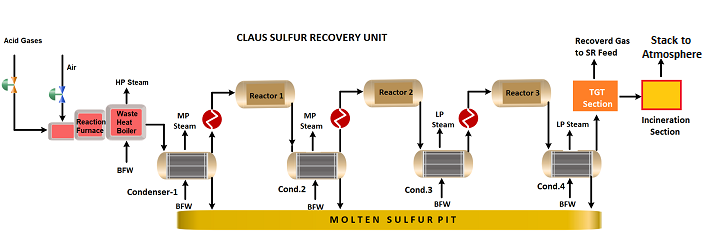
Sulfur Recovery Section
- In the Claus Sulfur Recovery Unit (SRU), the H2S-rich feed gas is mixed with air for partial combustion of the H2S to produce a 2:1 molar ratio of H2S to SO2. This reaction is highly exothermic. Claus process operating conditions are a high-temperature (~1000 °C), and low-pressure ( 21 PSIG/1.5 Barg) in a reaction furnace with a stoichiometric amount of air. The Claus process reaction equation is as follows;
6H2S (g)+ 3O2(g) → 2SO2 (g)+2H2O (g) + 4H2S (g)
- The H2S and SO2 now react to form sulfur via the Claus reaction. By the end of the reaction furnace, about two-thirds of the original H2S has been converted to sulfur. The temperature is 1,830 F (1,000 °C) at the reaction furnace outlet.
2H2S (g)+ SO2 (g) → 3 S(g) + 2 H2O (g)
- The reaction furnace fluids are cooled in a waste heat boiler and steam generator to condense the sulfur formed. The sulfur is removed from the steam generator as a liquid through a steam-jacketed seal leg or sulfur trap.
- The remaining gases are reheated by a high-pressure, superheated steam exchanger or an in-line fired reheater and then pass through a catalyst bed that is primarily alumina. The pressure is still low and the temperature is 580 F (305 C). The alumina catalyzes the Claus reaction, converting about two-thirds of the remaining H2S/SO2 to sulfur. The reactor effluent is again cooled. The sulfur is condensed and removed. Overall, about 89 % of the original H2S has been recovered as liquid sulfur.
- Step 4 is usually repeated twice more, at progressively lower temperatures 437 °F & 392 °F (225 & 200 °C). The overall recovery at the end of this “3-stage Claus” configuration would be about 98~99 %. The sulfur condenser for each subsequent stage operates at slightly lower temperatures but is still in a range to keep sulfur molten.
- The condensed, molten sulfur from the Claus stages is collected, usually in an enclosed sulfur pit, and maintained at about 285 °F (140 °C) using steam coils. The vapour space of the pit may be vented at a high point through a steam-jacketed vent or it may be swept by air or inert gas.
Tail Gas Treatment Section
- The Claus tail gas after processing can often be sent to an incinerator and emitted into the atmosphere in many locations. After the 3-Claus catalytic stages, the recovery would be close to 99 % of the sulfur. But for recovery ~99.9 %, the remaining sulfur is also recovered and recycled back to SRU feed.
- The tails gas leaving the Claus plant has a 2:1 molar ratio of H2S: SO2 with a few traces of other sulfur species and elemental sulfur vapour. The absorption system can work on H2S, so in the first step, the tail gas is hydrogenated of all sulfur compounds to H2S.
- The tail gas is heated to ~ 570 °F (300 °C) and enters the hydrogenation reactor. The reactor contains a cobalt-molybdenum catalyst which promotes the reduction of all oxidized sulfur compounds to H2S.
- The hydrogenation reactor effluent is cooled in an exchanger followed by a quench tower. In some processes, the quench tower has a caustic scrubbing section to absorb any trace SO2 that remains, since this can degrade the absorber solution. The gas leaving the quench and cooling system will be around 100~130 °F (50~90 °C).
- The cool gases enter a trayed or packed absorber where the H2S is scrubbed out of the gases using a lean amine solution. The rich absorber solution is sent to a regenerator system. The H2S released is recycled back to the Claus SR unit to join the incoming feed acid gas.

Incineration Section
The clean absorber gas can have less than 10 ppm of total sulfur compounds remaining after the Tail Gas Treatment Section. It is then sent to a final thermal Incinerator to convert gases such as H2S, COS, CS2, and elemental sulfur to SO2 by complete oxidation with excess air for a safer emission at the final stack. Increasing temperature up to ~ 760°C can achieve residual H2S content of not more than 10 ppm.
For further information, discussion and queries please comment in the box below or contact us at admin@ or follow us on Facebook & LinkedIn.
Certified Functional Safety Professional (FSP, TÜV SÜD Germany), Certified HAZOP & PHA Facilitator, LOPA Practitioner, QRA/FERA and Specialist in SIL Verification & Functional Safety Lifecycle, with 18 years of professional experience in Plant Process Safety across Petroleum Refining and Fertilizer Complexes.
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain