Sulfiding, pre-sulfiding, or pre-sulfurizing is the activation process of hydrotreating catalysts that converts catalyst metal oxides into metal sulfides by reacting with H2S (Hydrogen Sulfide) in the presence of hydrogen gas. The sulfided metal form is the most active state of the hydrotreating catalyst and will facilitate the hydrodesulfurization reactions.
The newly manufactured or regenerated catalysts are in the oxide state which is an inactive condition of the catalyst metals. They are converted into sulfide form, which is the active state of the hydrotreating catalysts. More about hydrotreating catalysts can be found in my previous blog “ Hydrotreating Catalysts”
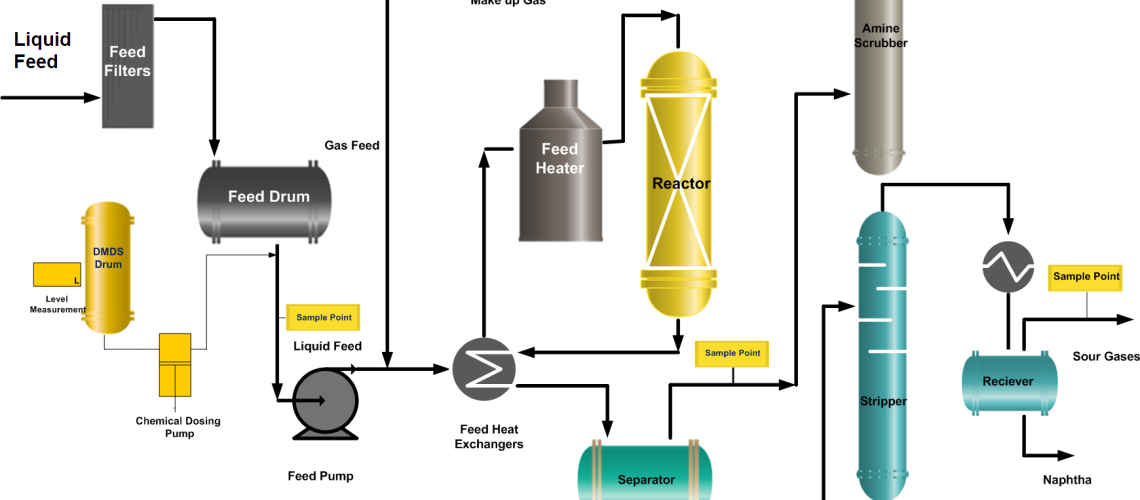
Most of the time the sulfidation process of the hydrodesulfurization catalysts is, in-situ (at the same site) but the trend is also rising to adopt the pre-sulfided (ex-situ sulfiding) catalysts. The ex-situ sulfiding has the advantage of less downtime, minimum effects of pollutants or toxic chemicals, and efficient sulfiding results.
Types of Sulfiding
The Sulfiding process of hydrotreating catalysts can be accomplished either in the vapor phase or liquid phase. Both of these sulfiding methods are commercially utilized but the wet phase sulfiding in the presence of an organic sulfiding compound is most preferred.
Dry Phase Sulfiding or Gas Phase Sulfiding also called vapor phase sulfiding is executed in the presence of hydrogen by injecting a chemical (dimethyl disulfide (DMDS) or dimethyl sulfide (DMS) which can easily be decomposed to H2S and no oil is used.
Wet Phase Sulfiding or Liquid Phase Sulfiding: Liquid-phase sulfiding can be carried out with or without the addition of organo-sulfur compounds in the feed. In the case, the organic compound is not to be utilized then the feedstock is generally a gas oil-type with sulfur in ranges 1~2 weight %. When the organic compound is used then the H2S necessary for the activation of the catalyst is generated by the decomposition of the sulfur compounds.
The preferred sulfiding method in the industry is a liquid phase with a sulfur-containing compound because it requires less time. Another advantage of the liquid phase over gas-phase sulfiding is that all the catalyst particles are fully wet thus reducing the chance of catalyst bed channeling which can occur if the catalyst particles are allowed to dry out.
Further, in wet or liquid phase sulfiding, the quantity of sulfiding agent can easily be manipulated as per requirement thus giving control over sulfiding. Moreover, in the wet method liquid serves as a heat carrier during the sulfiding exothermic reactions and saves the catalyst from pre-mature coking.
The modern Type-II catalysts require the wet sulfiding method because of the presence of a chelating agents. These chelating agent compounds in the presence of startup oil help to convert the active metals into multilayers.
In the case of ex situ presulfurization of the catalyst, sulfur compounds are loaded onto the catalyst. The activation occurs when the catalyst, which has been loaded in the reactor, is heated up in the presence of hydrogen and the sulfur compounds decompose to H2S. During this period, the H2S scrubber is offline so that the H2S can accumulate in the recycle gas circuit.
Catalyst Sulfiding Reaction
At a temperature above 205 °C, the DMDS is decomposed into H2S and then facilitates the sulfiding reactions in the presence of Hydrogen as follows;
MoO3 +2H2S+H2 → MoS2+ 3H2O
Co8O9+8H2S +H2 → 8CoS + 9H2O
NiO2+H2S+ H2 → NiS+ 2H2O
The Sulfiding Agents
Many Sulfding agents are used in the market which includes DMDS (Dimethyl Disulfide), DMS (Dimethyl Sulfide), TBPS (Tertiary-butyl Polysulfide), TNPS (Di-tertiary nonylpolsulfide), etc. But sulfiding agent DMDS is widely used around the globe due to its high H2S content i.e. 68% as compared to other agents.
The required quantity of chemicals is determined by the amount of catalyst metal oxide. Its required quantity is calculated on a stoichiometric basis then the actual amount is maintained more than the theoretical amount to ensure completion. Please view the post for stoichiometric calculations of DMDS required for Sulfiding.

Sulfiding Procedure
It is extremely important to follow the supplier’s instructions regarding the presulphiding procedure, specially referred to rates of temperature and flows, in order to assure complete activation of the catalyst and to avoid coking or undesirable reduction of metals. Every catalyst provider has a specific procedure of sulfiding to achieve the maximum efficiency of the catalyst. Sulfiding services can also be hired to perform in situ sulfiding or ex-situ sulfiding. These are only general steps, so please follow specifically your vendor procedures and instructions.
1. Air Freeing , Nitrogen Purging and Leak Testing
Before starting the sulfiding of the catalyst, the reactor circuit is air freed, and leak tested by Nitrogen purging and pulling vacuum. The oxygen content is reduced to less than 1 %. The leaky points should be rectified before introducing hydrogen gas.
Further, the downstream fractionation section of the hydroprocessing unit should be flushed with flushing oil to remove any debris due to maintenance activities.
2. Catalyst Dry Out
Before sulfiding, the catalyst is dried out to remove moisture content trapped in the catalyst and any neutralization soda or water present in the equipment due to maintenance activities. Catalyst dry out is performed by circulating hydrogen gas through the catalyst at maximum capacity of the recycle gas compressor. Recycle gas sweep any water present in the system. Dry out is enhanced, by increasing temperature and pressure of the catalyst.
The critical step of catalyst dry out is the increase the reactor temperatures and achieve the MPT (Minimum Pressurization Temperature) and increase the reactor pressure up to normal operation pressure. The maximum dry out of catalyst is achieved at high pressures.
The biggest challenge for achieving the normal pressure is the reactor skin temperatures due to MPT limitation but can be achieved easily if the fresh catalyst temperatures can be increased up to a safe point or MPT requirement is lower skin temperatures.
Temperature of the catalyst is increased up to 100~120 °C while pressure is increased up to normal pressure. Water is removed from the boot of the separator and low point drains at the reactor circuit. Dry out step should be continued until water stops appearing in the high-pressure separator.
During dry out step, care must be taken to control the catalyst temperatures below the maximum temperature limit (~130 °C) of the fresh catalyst, otherwise it can damage the catalyst.
3. Pre-wetting Step
Pre-wetting is carried out to soak the catalyst with oil and remove the debris or dust produced during loading. Oil is introduced while increasing the reactor temperature to ~ 100 °C, depending on the designer’s guidelines. The oil used for the whole process should be straight run and with designed specifications. After oil introduction, the heat of absorption will produce, and care should be taken to avoid sudden temperature rise. The heat of absorption is a one-time phenomenon and the temperature drops within minutes after the heatwave passes.
Catalyst wetting with feed is important for in-situ sulfiding performance, preferably at higher liquid rates (~ 50 % of design flow rate) for good flow distribution. Initial oil introduction at full rate (50 %) is helpful to avoid channeling and control the exotherms due to heat of adsorption. Complete catalyst wetting allows this heat of absorption to essentially pass through the unit before beginning the sulfiding process.
Prewetting step normally continued for 4~6 hrs, and reactor to fractionator section is flushed with oil with once through circulation. Once through circulation is stopped when no particles are observed from the oil at the unit out. Close circulation of oil is required during sulfiding. In close, circulation, the oil is recycled to feed surge drum from the fractionation section. The recycle route should contain feed filtration system so that any particles in the oil stream can be removed before going to the reactor.
Before, pre-wetting the fractionation or stripping section of the hydrotreating units are normalized by pressurization and flushing with oil. Once through, circulation to slope is maintained until, clear oil is observed from the unit outlet.
4. First Phase Sulfiding at Low Temperature
After completion of the pre-wetting step, the reactor temperatures are increased to the decomposition temperature of the sulfiding agent I.e. 200~230 °C in the case of DMDS. The temperature rate should not be more than the recommended, normally 15~20 °C per hour. Total sulfur content (including Sulfur in HC & Sulfiding agent) in the feed should be 1.5~2.0%.
As sulfiding is an exothermic and consume hydrogen, reactor temperatures should be controlled within limit (235 °C) by regulating the DMDS dosing rate. Initially, the DMDS dosing rate should be minimum to avoid high exotherms.
Monitor H2S concentration in the recycle gas at regular intervals. Hydrogen Sulfide breakthrough will be observed if H2S concentration has increased abruptly in the recycle gas.
During, sulfding water is produced and appeared in the boot of high-pressure separator. The water produced during sulfiding can be as much as 10 % of the catalyst weight. Monitor the boot of high pressure separator and drain the water as necessary.
The minimum hydrogen purity in Recycle gas is ~ 70 %. As, during sulfiding there is no bleed gas from the system and Amine Scrubber is also not service, recycle gas purity gradually decreases. Therefore, it is better to charge high purity make up gas. Make up gas, is added to maintain the system pressure. Maintaining an adequate hydrogen level reduces the time required to complete sulphiding and also prevents premature coking in the catalyst.
Furthermore, most part of sulfiding (~70%) is completed in the low temperature step is shown by the consumption of DMDS.
5. Second Phase Sulfiding at High Temperature
This step is performed at 315~345 °C while maintaining the injection rate i.e. 1.5~2.0 %. Initially with the increase of reactor temperature, H2S content in the recycle gas will reduce below 1000 ppm, and then after 4~5 hours, H2S will rise to approximately the same as added. During this phase, H2S content is maintained in Recycle Gas at approximately 3500-4000 ppm.
The step will be considered complete when water in High-Pressure Separator stops appearing and there is no further uptake of H2S or sulfur. After achieving high temperatures, the conditions are held for 4 hours, then stop DMDS injection. Some, service providers recommend to continue DMDS until the feed replacement with the fresh straight run feed.
Sulfiding is considered complete if 1. no further water production and appearance in the High-pressure separator boot, 2. no further Hydrogen consumption, due to no sulfiding reactions, 3. no significant temperature rise on the catalyst as sulfiding is completed and no exothermic reactions and 4. DMDS consumption is 20~30 % more than the theoretical.
6. Unit Normalization
After completion of sulfiding of the hydrotreating catalyst, gradually the fresh feed is introduced, the fractionator bottom is diverted to slop and close circulation is stopped. It can take to 4~6 hours, to replaced the feed with fresh straight feed. Initially, rate of fresh feed should be low to observe the behavior of the catalyst with fresh feed.
During feed replacement start wash water injection in the reactor circuit and establish Amine circulation for recycle gas scrubbing. Also, set reactor quenched control valves on auto to control the interbed temperatures.
Sulfiding Monitoring
The following parameters should be monitored at specific intervals, normally after an hour;
1. Injection Rate
The dimethyl Disulfide sulfiding agent injection rate should be monitored closely to find the actual consumption in comparison to the theoretical. This is calculated by measuring the level reduction of the DMDS measuring drum.
2. Recycle Gas Purity
Hydrogen purity is continuously monitored using an online analyzer and is normally more than 80% in the Sulfiding process. H2S content in the recycle gas is monitored using the Dragger tube or by some other method.
3. Reactor Temperature
The reactor temperature is a very critical parameter and uncontrolled rise may lead to premature coking of the new catalyst. So, temperature-rising rates are maintained at the recommended. In case of any sudden rise in the reactor temperature at any stage during sulfiding control the temperature by reducing reactor inlet temperatures, using quench gas and also reducing DMDS injection.
4. Water content in the Separator
Sour water is the by-product of the Sulfiding process if water continues to come then Sulfiding is in progress. The separator boot level is noted down at regular intervals.

Troubleshooting of the Sulfiding Process
- To maintain the activity of the chelated hydrotreating catalyst during the dry-out process, avoid exposing it to high temperatures (~135°C) until pre-wetting begins. Allow sufficient time for the catalyst pores to be thoroughly soaked with oil before breaking down the chelating agent, which decomposes at 140~150°C, producing water as a byproduct.
- During pre-wetting step, quickly introduce the feed in the reactor and increase the feed up to 50% within one minute. This initial feed rate will help to control the heat release due to oil adsorption and help to avoid channeling in the catalyst. The initial feed rate can be 50~80%, recommended by the catalyst provider.
- When oil flows over the dry catalyst, a significant heat wave can form as the oil absorbs onto the catalyst. This heat wave typically ranges from 5~15°C but can reach up to ~30°C in certain areas. During this phase, closely monitor catalyst temperatures and efficiently introduce quench gas before the heat wave reaches each lower catalyst bed, indicated by a sudden temperature rise at the outlet of the bed above.
- If an exotherm exceeding 28°C is observed, maintain or increase the feed rate and reduce the feed temperature. However, ensure the feed temperature is not reduced to the extent that the reactor skin temperature falls below the Minimum Pressurization Temperature (MPT).
- Catalysts at normal ambient conditions can contain a moisture content of about 2~5% because of their hygroscopic nature. If this is still more than 1 %, it will restrict the sulfiding reaction. Further, at high temperatures it will convert into steam, causing physical damage to the catalyst. So, ensure that the maximum amount of water has been removed before sulfiding.
- The oil used for sulfiding should be straight run (SR) so that there are no olefins or other material that could lead to coking. The oil should preferably have a final boiling point (FBP) of less than 670°F (355°C) while the final temperature should be in the 600~650°F (315~344°C) range, typically depending on the unit’s capability.
- During the sulfiding of the catalyst, if the dosing rate is higher and the catalyst temperature is lower then suddenly exotherms may appear when the catalyst reaches the decomposition temperature of the sulfiding agent. This high temperature may hammer the catalyst activity due to coking.
- More than half of the sulfur should be consumed during the low temperature sulfiding step. Consumption, less than the estimated quantity, is an indication there may be some problems with bypassing the reactor or maldistribution of the feed.
- The heating rate should not be more than the design, because a sudden increase in temperature may lead to coking. If a sudden temperature rise is observed at any stage then adjust or in extreme cases stop firing the heater. Use quench gas to control the interbed temperatures and reduce the DMDS dosing rate as per conditions of the catalyst.
- The catalyst should never be left for extended periods in hydrogen environment at temperatures and pressures greater than 450 °F (232°C) and 435 psig (~30 Bar) respectively, as there is a potential of reduction of catalyst metals (i.e.,Ni, Co and Mo). The Ni or Co will go into the metallic state while the Mo will form MoO2. These resulting materials will not sulfide, so there will be loss of significant portion of the active metals in the catalyst. Temperature should be reduced if there is a need to shutdown or put the startup on hold for any given length of time due to mechanical issues. This helps avoid possible metals reduction and/or coking and subsequent catalyst deactivation.
- When there is a deficient atmosphere of H2S at a high temperature (greater than 200 °C ) in the presence of hydrogen, oxygen will convert to water and resulting in the reduction of catalyst metal. Reduction results in permanent activity loss of the catalyst.
- DMDS decomposition and the sulfiding reactions are both exothermic, hence heatwave will generate across the catalyst. Also, high heating rates will cause premature coking of the catalyst. In case of unexpected exotherms immediately hold your step or cut out the heat source until conditions normalized.
- To avoid hydrogen embrittlement of the reactor, pressure should never exceed the metallurgical limits. Besides, ensure minimum pressurization temperature before going above the critical pressure as advised by the manufacturer.
- After the completion of sulfiding, the catalyst is super active, and cracked stock may lay down the permanent scale or coke on the catalyst. Hence, after sulfiding for at least three days the cracked stock should not be used then gradually introduced. Less reactive or straight-run oil will produce small soft coke on the catalyst surface and will decrease the super activity.
- It is important to avoid injection of DMDS at a high rate early before the low temperature stage of sulfiding. Over injection at low temperatures can lead to sudden exothermic reactions over the catalyst as the temperature reaches to the decomposition stage of the DMDS with potential damage to catalyst. Further, excessive recycle gas density and low hydrogen purity, will cause the need to purge sour gas from the system to maintain the purity and add make up hydrogen in order to avoid catalyst damage due resulting from low hydrogen partial pressure. The DMDS injection should be started above 200 °C of the catalyst.
- During sulfiding, recycle gas should not be bleed from the system to maintain the H2S in the system. System pressure can be maintained by using fresh makeup hydrogen. Also, the gas treating system i.e. Amine Scrubbing should be stopped.
- The last and most important point is about safety, as sulfiding agents contain a very high content of H2S, low flash point, ignition temperature, and are highly explosive. Strictly, follow the MSDS instructions and train the staff about the hazardous nature of the sulfiding agent. Also, take special care while sampling and use live breathing apparatus or some other method to avoid H2S exposure.
Top References
- Hydroprocessing for Clean Energy by Frank (Xin X), ZHU, Richarg Hoehn, Vasant Thakkar, Edwin Yuh
- Safer and Smarter Sulfiding of Hydrotreating Catalysts
- www.reactor-resources.com
- American Fuel and Petrochemical Manufacturers
- www.digitalrefining.com
For further information, discussion and queries please comment in the box below or contact us at nasir.hussain@ or follow us on wwwFacebook & LinkedIn.
Certified Functional Safety Professional (FSP, TÜV SÜD Germany), Certified HAZOP & PHA Facilitator, LOPA Practitioner, QRA/FERA and Specialist in SIL Verification & Functional Safety Lifecycle, with 18 years of professional experience in Plant Process Safety across Petroleum Refining and Fertilizer Complexes.
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain






9 thoughts on “Sulfiding of Hydrotreating Catalysts and its Troubleshooting”
Good man
Dear Mr.Nasser,
how we can calculate the amount of DMDS is required to complete the sulfiding?
Thanks for sharing very good practical and theoretical information.
Good morning,
what happen if the hydrogen is lower than target value during the Sulfiding Procedure? And what if it upper?
Thanks a lot
High purity of hydrogen gas means fast reactions of sulfiding and more water is produced in the boot as a result sulfiding will be completed in less time. But during high-purity gas, H2S should remain sufficiently high as recommended otherwise catalyst reduction reactions may proceed.
Low-purity hydrogen gas will slow the sulfiding reactions.
What’s the minimum H2S is required in the recycle gas after sulfiding is completed and feed cut in not started due to leak of hydrogen in HP loop and this leak required to shutdown the unit and purge with nitrogen?
Once the sulfiding has been completed, there might be need to maintain some fixed amount of H2S in the recycle gas. Nearly 10 ppm H2S would require but contact with your service provider for guideline.