The primary function of Hydrotreating catalysts is to remove sulfur, nitrogen and other contaminants from the refinery feedstocks. In addition, they improve product properties by adding hydrogen and in some cases improve the performance of downstream catalysts and processes. The hydrotreating catalysts are high surface area materials consisting of an active metal component and a promoter, uniformly dispersed over a support material. The type of catalyst required for hydrotreating service depends upon the feedstock properties and product quality requirements.
The most commonly catalyst metals used in hydrotreating are Mo (Molybdenum) and Tungsten promoted by Ni (Nickel) and Co (Cobalt) because of their high activity, selectivity, and stability. Tungsten (W) is less preferred because of its high price and is applied in hydrocracking service.
The catalyst metals are impregnated as a thin layer on the support material that provides a wide surface area with the small weight of the catalyst. Support material must have high mechanical strength and thermal stability in the reaction environment. The most widely used support material is Alumina because of its high mechanical strength and low cost. In addition, Silica (SiO2), Magnesia (MgO), Zirconia (ZrO2), and zeolites are also used.
The newly manufactured catalysts are in the oxidized state and are sulfided to convert into an active form. For sulfiding of hydrotreating catalysts please view my previous blog.
Reactions selectivity of the catalysts in the hydrogen environment and petroleum feedstock is shown in the table below. Co-Mo requires less Hydrogen consumption and shows excellent Desulfurization activity and low Denitrogenation activity. In the contrast, NiMo requires high hydrogen consumption and is utilized for heavier feeds to remove Nitrogen, Oxygen, Metals, and aromatics saturation.

Hydrotreating Catalysts Composition:
Hydrotreating catalysts composition has varying amounts of active metals and promotors depending upon the specific application. In general, they contain up to 26% active metal and 25 % promotor components.

Properties of Hydrotreating Catalysts:
Physical properties of the particles play a significant role in the activity of the catalyst. The catalyst provides high surface area and pore size that have the ability i. to catch scales, coke, and metals, ii. to withstand coke formation, iii. to withstand high pressure and temperature conditions, and iv. to support the required hydrotreating reactions, for the expected operating cycle.

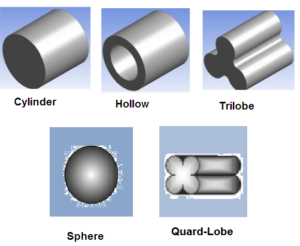
Shapes of Hydrotreating Catalysts:
The hydrotreating catalysts come in various shapes and sizes, depending upon the application service and manufacturer. Common shapes include cylinder, sphere, hollow ring, tri-lobe, quad-lobe, and pellets.
Type I and Type II Hydrotreating Catalysts:
Type-I and Type-II Catalysts represent the nature of the bonding between the active phase of catalyst and support. The active phase of catalyst is sulfided form i.e.CoMoS, NiMoS.
|
Type I Catalyst |
Type II Catalyst |
| A mainly single layer of the active phase | Consists of multiple layers of the active phase |
| Incomplete or partially sulfided catalyst | Fully sulfided catalyst |
| Strongly bonded with Alumina support (Mo-O-Al) | No bonding with Alumina support. Only weak force dipole dipole interaction exists |
| Poor access to active sites | High access to active sites |
| Low hydrogenation activity | High Hydrogenation activity |
| Less active than Type II | Highly active than Type I |
Unsupported Transition Metal Catalysts: The significant improvement in hydrotreating catalyst activity can be observed in the unsupported transition metals catalyst. Unsupported catalysts consist of only active phases and are 50~100% more active than Type II catalysts.
Some highly active commercial-grade hydrotreating catalysts are as follow;
- HALDOR TOPOSOE’S, TK-611 HyBRIM catalyst
- ALBEMARLE’S, Nebula catalyst
- ART Hydroprocessing, SmART Catalysts Systems
- UOP UltiMet™ High-Activity Catalyst
Hydrotreating Catalysts Suppliers:
There are various suppliers of hydrotreating catalysts world wide, which provide high quality hydrotreating catalysts. Some if these include Haldor Topsoe, Axens, ART Hydro processing, Shell, Albemarle, UOP Honeywell, Johnson Matthey.
For further details, discussion, please comment in the box below or contact at admin@
Certified Functional Safety Professional (FSP, TÜV SÜD Germany), Certified HAZOP & PHA Facilitator, LOPA Practitioner, QRA/FERA and Specialist in SIL Verification & Functional Safety Lifecycle, with 18 years of professional experience in Plant Process Safety across Petroleum Refining and Fertilizer Complexes.
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain







12 thoughts on “Hydrotreating Catalysts”
Dear Nasir,
Thank you for the blog. This is a very detailed level and informative blog I found for somebody like me who is new to this category.
As I am working on a research paper, I would like to understand more about the process of manufacturing the catalyst, be it used for Naphtha Hydrotreating for gasoline, Diesel/ Kerosene/Cracked Naphtha, etc.
Also, I am keen to know, what is the cost incurred in manufacturing X tonnes of catalyst, like – Raw material Labor, utilities, overheads, etc (roughly per cent).
As these catalysts remain active for a certain period of time, what could be their average life span? And after the useful life, how are they disposed of? Is there any cost associated in disposing catalyst?
Looking forward to hearing from you.
Hydrotreating catalysts are metals impregnated on support.
Step-1: There are two methods for Alumina support formation, 1. Oil dropping method 2. Support preparation by extrusion.
Step-II: Three methods of catalyst manufacturing are used on commercial scale, 1. Dry or wet impregnation, 2. Co-mulling 3. Hot soaking/dip impregnation. Simple catalyst manufacturing process can be seen on the link below.
https://cbe.rutgers.edu/catalyst-manufacturing-center
Average life of hydrotreating catalysts is ~3 years but in less server operations can be extended up to 10 years.
If the catalyst has been in severe operating conditions and irreversible poisons had attacked, then it is unlikely to regenerate the catalyst. If the catalyst has been deactivated due to coking during normal operation, then it is preferable to regenerate the catalyst to save money and environment. Spent catalyst that cannot be regenerated, can be reuse for a different catalytic or chemical process. If no other option left, then valuable metals should be recovered before disposal to landfill. Landfill is always less preferred because of its long term health and environmental hazards.
There are number of options available to treat the spent catalyst.
• Regeneration, rejuvenation to reuse in the same process
• After treatment or regeneration use of the catalyst in some other process.
• Recovery of the valuable metals from the spent catalyst
All of the above methods involve cost.
Very nice effort , may Allah provide success