The acid alkylation process that converts low molecular weight streams into alkylate, high octane, and gasoline blending components in the presence of strong Hydroflouric Acid is called a Hydrofluoric Acid Alkylation Unit (HFAU). The hydrofluoric acid alkylation process only makes a small number of organofluoride side products that are removed from the reactor continuously and the used Hydroflouric acid is replenished. HF alky units are also capable of processing a broad range of light-end feedstock with propylene and butylene and produce high octane rating alkylate, as compared to sulfuric plants. Hydrochloric Acid is dangerous and extra precautions are taken to ensure safe unit operation.
The hydrofluoric acid alkylation Process has the following advantages over Sulfuric Acid Alkylation Unit (SAAU) Process;
- The low viscosity of Hydroflouric Acid and high solubility of isobutane in the acid allows a simpler reactor design.
- The hydrofluoric acid alkylation Process process has no mechanical stirring as in the SAAU process.
- Less sensitive to temperature so that cooling water can be used as a coolant instead of refrigeration.
- Hydroflouric acid consumption is low due to the nearly complete regeneration of HF acid also disposal of acid is not necessary.
- The flexibility of operations in temperature and isobutane-to-olefin ratio and less need for turbulence or agitation.
Two hydrofluoric acid alkylation processes (HFAU) are commonly available. These are the Phillip process and the UOP process. A Phillips design eliminates the use of an acid circulation pump, its seal, and many associated flanges due to the density difference between LPG and acid phases. Many gravity-circulated units were licensed as the scheme permitted the construction of the reactor, cooler, and settler as a single pressure vessel.
After UOP acquired the ConocoPhillips alkylation technology, an improved design called the AlkyPlus process was developed to exploit the best commercial features of each heritage technology. The resulting process design demonstrates lower risk and best design when compared to either of the parent technologies.
Process Description of Hydrofluoric Acid Alkylation Unit
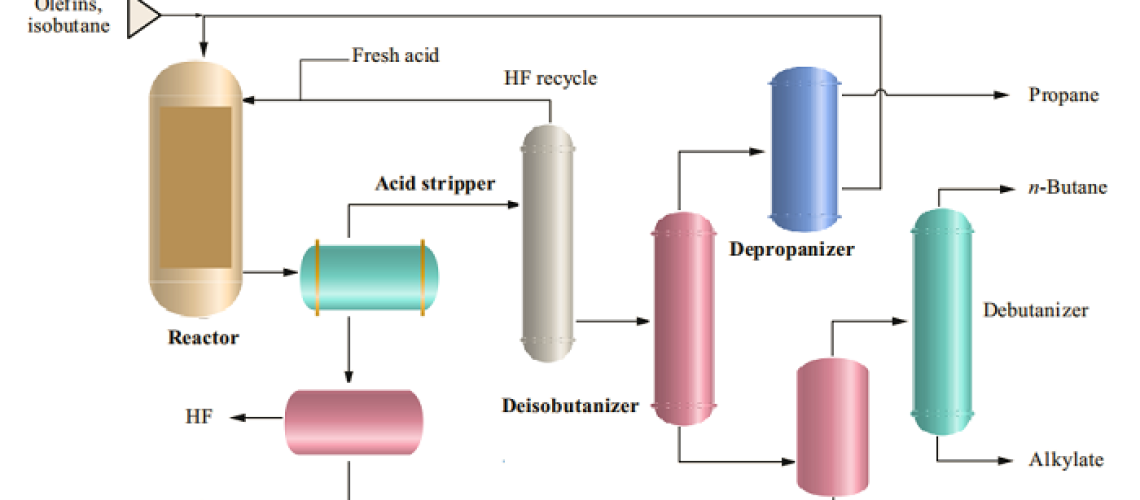
The process Flow Diagram of the Hydroflouric Acid Alkylation Unit is shown below;

1. Feed Pre-treatment Section
Olefins and isobutane products from Hydrocracker, FCC, Coker, reformer, isomerization, and Crude distillation units are de-euthanized, treated with amine, and applied Caustic Extraction (Merox Unit) to remove H2S and Mercaptans and dried. Acetone, ethers, and alcohols are also removed through MTBE or the Selective Hydrogenation process.
2. Reaction Section
After feed pretreatment, the olefin, isobutanes, and recycled isobutane are injected into the water-cooled HF reactor with a circulating HF acid catalyst. The molar ratio, of isobutane to olefin is in the range ~ 6~14. The cooling water flows through the reactor tubes, removes the heat of the reactor from highly exothermic HF alkylation reactions, and maintains the reaction temperature at 16 ~ 38°C (60 to 100°F). The residence time in the HF reactor is 20~40 seconds and the alkylation reactions are very fast with 100% Olefin saturation.
The excess isobutane, alkylate product, nonreactive hydrocarbons (propane, n-butane), and the HF acid catalyst pass on to the settler vessel. The dense acid phase separates from the hydrocarbons rapidly by gravity and is then pumped or circulated based on gravity differences back to the reactor. The hydrocarbons containing dissolved HF flow from the settler to the isostripper.
3. Fractionation Section
The HFAU Fractionation section typically consists of an iso-stripper, a depropanizer, and an
HF stripper. The iso-stripper is the main tower that strips HF overhead, separates the isobutane recycle stream, and produces n-butane and alkylate product streams. Alkylate is drawn off the bottom of the tower, cooled in exchangers, and sent to product storage. The next product to draw up the tower is the n-butane side draw and above that is the large isobutane recycle draw. On most UOP units, the isobutane draw is located below the feed tray to minimize HF in the isobutane recycle.
The isostripper overhead vapor is a propane-enriched isobutane stream that contains HF, and this stream is condensed and separated in a settling drum. The heavier HF phase is returned to the reactor section. The hydrocarbon phase is charged to the depropanizer.
In the same way, the depropanizer overhead contains propane products and HF. The overhead vapor is condensed and the two liquid phases are separated in the overhead receiver. The heavier acid phase is returned to the reactor section, and the acid-saturated hydrocarbon phase is fed to the HF stripper column where the HF is stripped overhead and joins the depropanizer overhead vapor where the HF can be recovered. The HF stripper bottom stream is an acid-free propane product that is treated with hot alumina to remove organic fluorides, cooled, and treated over a bed of granular KOH to remove traces of HF and water.
4. Acid Regeneration
A key advantage of HF alkylation over sulfuric alkylation is the ability to recover the acid from the by-product polymer, water, and other contaminants. A small stream of circulating acid is stripped with superheated isobutane in a small Monel tower called the acid regenerator or acid rerun tower. The regenerator overhead is HF and isobutane that are recycled to the reactor; the regenerator bottom is the polymer and the HF-water azeotrope, is neutralized with aqueous NaOH or KOH. The neutralized polymer has good fuel value.
Process Variables in HF Alkylation
Key process variables of the HF Alkylation process are as follows;
Reaction Temperature
Reaction temperature has a significant influence on the octane number of the alkylate product. Almost all HF alkylation reactors are operated below 110 °F. Above 120 °F polymerization and cracking side reactions become excessive, reducing alkylate quality and octane number, and minimizing acid regeneration capacity of the HF alkylation unit would not be able to maintain the acid strength.
Isobutane to Olefin Ratio
As the isobutane to olefin ratio is increased, alkylate octane number increases, and the polymer production decreases. Therefore, the flow of isobutane recycle stream is usually kept at maximum according to the capacity of the isostripper.
Acid Strength
Acid strength is usually kept at 85~95 wight % Hydroflouric acid. Fresh acid is supplied by acid manufacturers at 99.97 wt% Hydroflouric acid. This purity is too high for the optimum performance of the alkylation process. The water content of the acid is typically controlled between 0.5~1.5 wt%. Water levels below 0.5 wt% cause a reduction in octane and water above 1.5 % lead to higher corrosion on the carbon steel piping and vessels. The optimum acid strength is maintained by a balance between the performance of the unit feed-treating systems for sulfur and water removal and acid regeneration operation.
Acid to Hydrocarbon Ratio
The acid to Hydrocarbon volume ratio used in the Hydroflouric acid alkylation process is generally 1:1 or higher. At some point, below 0.8:1.0, excess polymerization occurs. In the most extreme cases, alkylate production could stop.
Pressure
Pressure does not have an effect on the process as long as it is kept high enough to prevent the vaporization of hydrocarbon and acid in the reactor section.
For further information, discussion and queries please comment in the box below or contact us at admin@ or follow us on Facebook & LinkedIn.
Certified Functional Safety Professional (FSP, TÜV SÜD), Certified HAZOP & PHA Leader, LOPA Practitioner, and Specialist in SIL Verification & Functional Safety Lifecycle, with 18 years of professional experience in Plant Operations and Process Safety across Petroleum Refining and Fertilizer Complexes.
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain