Liquefied petroleum gas or LPG is the term applied to the propane and butane hydrocarbons that exist in the gaseous state under atmospheric ambient conditions but can be converted to the liquid state under conditions of moderate pressure at ambient temperature. A commercial LPG mixture may also contain ethane and ethylene as well as a volatile mercaptan. LPG is a valuable fuel that is used worldwide as an alternative fuel for various applications; residential, commercial, agribusiness, industrial, and autogas.
Most of the world supply of LPG comes from the wet natural gas processing fields, and the remaining ~ 40% of the LPG is produced in oil refineries from crude distillation Unit, naphtha reforming units, fluid catalytic cracking units (FCCUs), delayed cokers, hydrocrackers, and other conversion processes. LPG produced in the gas fields or petroleum oil refinery may be treated with amine or applied Benfield process to remove Acid gases. In the Benfield, process gases are treated with a solution of potassium carbonate containing some additives. In the oil refinery, after amine treatment, the LPG is further treated in the MEROX unit.
LPG is a relatively clean-burning fuel that produces less greenhouse gas emissions throughout the whole fuel cycle than any other fossil fuel. The absence of sulfur and very low levels of nitrogen oxides (NOx) and particulate emissions during its combustion make LPG a most environmentally friendly source of energy.
Uses of LPG
- LPG is the most adaptable fuel used in domestic applications. LPG is used for cooking, and heating as well as in a large number of appliances, such as ovens, stovetops, and refrigerators in homes, hotels, and restaurants.
- LPG is increasingly being used as automobile fuel because of its low cost compared to gasoline and diesel.
- In industry, LPG is used to power industrial ovens, kilns, furnaces, and various process heating applications. LPG is used in brick kilns and aluminum die casting, in ceramics, and in glass manufacture. LPG is used to heat bitumen for road building.
- In agriculture, crop drying, waste incarnation, greenhouse heating, and running power equipment.
- As a feedstock for chemical manufacture, in water desalination plants, and in aerosol manufacture as a propellant.
Properties of LPG
Following are the main physical and chemical characteristics of Liquefied Petroleum Gas.
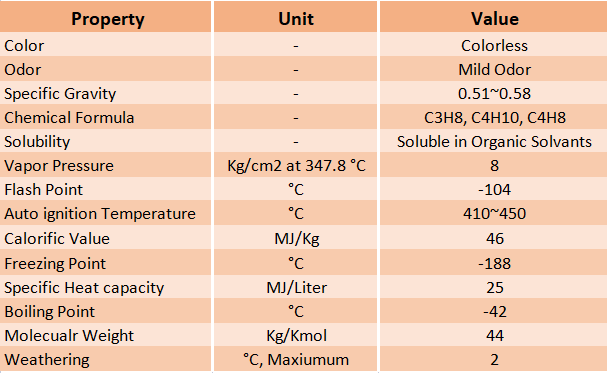
1. Colour
Both the liquid and vapor phases of LPG are colorless. During a leak, the vaporization of liquid cools the atmosphere. It condenses the water vapor to generate a whitish fog or icing, which may enable an escape of LPG to be seen.
2. Specific Gravity
LPG is a gas that is 1.5 to 2.0 times heavier than air at atmospheric pressure and temperature. At 15 °C, liquid LPG has a specific gravity of 0.51 to 0.58. Since LPG’s vapors are heavier than air, they will settle at ground level or in low-lying areas.
3. Chemical Formula
The normal constituents of LPG are propane (C3H8), propylene (C3H6), butane (C4H10), and butylenes (C4H8).
4. Solubility
At 20 °C, the solubility of LPG in water is less than 200 ppm (parts per million). LPG dissolves in organic solvents such as alcohol.
5. Vapour Pressure
At 37.8 °C, the vapor pressure of LPG is ~8.0 kg/cm2. Inside the LPG storage vessel, the pressure is equal to the vapor pressure corresponding to the temperature of the LPG in the storage vessel. The vapor pressure is affected by temperature and the hydrocarbon mixture ratio.
6. Odour
LPG has a very mild smell; hence it is required to add an odorant so that any gas that escapes can be immediately recognized. For this purpose, ethyl mercaptan is typically utilized as a stanching agent. According to IS: 4576, the amount to be applied must be enough to allow the detection of 1/5 of the lower limit of flammability or level 2 odor in the atmosphere.
7. Flash Point
The flash point of LPG is -104.4°C. The auto-ignition temperature for propane is 46.1°C and 405°C for butane. The auto-ignition temperature of LPG is around 410-580°C. Hence, LPG does not ignite spontaneously at an average temperature.
8. Calorific Value
Depending on the composition of the LPG, the specific calorific value is approximately 46 MJ/kg or 12.78 kWh/kg.
9. Freezing Point
The freezing point of LPG is -188 °C (-306.4 °F). As a result, while frost or ice may build on an LPG system, the LPG-propane itself does not freeze since the freezing point of LPG is so low. So, LPG does not freeze in the winter because the temperature outside of a laboratory is never that low.
10. Toxicity
While LPG is not harmful in the vapor phase, despite being slightly toxic, it can suffocate when present in high concentrations because it displaces oxygen. As a result, the vapor has minor anesthetic qualities.
11. Combustion
In addition to generating heat, the LPG combustion reaction increases the volume of the products. For complete combustion, LPG needs up to 50 times its amount of air. Therefore, it’s essential to have enough ventilation when burning LPG in enclosed spaces to prevent asphyxiation from oxygen depletion as well as the production of carbon dioxide.
12. Specific Heat Capacity
LPG has an energy content of about 25MJ per liter. One gallon of propane has 91,547 BTU (60°F) of LPG energy. 25MJ is also equivalent to 6.9kWh.
13. Boiling Point
At -42°C or -44°F, LPG (propane) begins to boil and turn into gas vapor. Due to the pressure that LPG is under in a gas cylinder, it remains liquid.
14. Flame Temperature
The Propane flame temperature is 1967°C (3573°F).
15. Molecular Weight
The molecular weight of LPG is 44.097 kg/kmol.
16. LPG Weathering or Volatility
The significance of the LPG weathering test is to find out the presence of heavier hydrocarbons in the LPG like pentanes and hexanes etc. In weathering test, 100 mL of liquid LPG sample is taken in a test tube and is allowed to evaporate. After 95 mL of liquid is vaporized, the remaining (5 ml) liquid temperature is noted. If the temperature is very low (below 0°C), then the proportion of propane in the mixture is more and if it is high (above 2°C), then the proportion of butane will be more. For domestic use, the maximum weathering temperature should be 2°C.
In the petroleum refinery, debutanizer columns, LPG weathering is controlled by reducing the top temperature of the tower, this will increase the LPG reflux to the tower. So, make a balance between Naphtha RVP and LPG weathering.
17. Moisture Content in LPG
The LPG should be dry and no free water should be present. The presence of moisture in LPG may be converted to ice during vaporization and may cause trouble in storage or piping. Pipelines that transfer LPG and the pumping equipment may be damaged due to the formation of ice.
Top References
- Liquified Petroleum Gas (LPG) topic wikipedia.com
- Fundamentals of Petroleum Refinery by M.A Fahim.
- Fundamentals of Petroleum and Petrochemical Engineering by Uttam Ray Chaudhuri
- LPG Physical Properties and Characteristics
- Liquefied Petroleum Gas- its Characteristics and Safety Requirements
For further information, discussion and queries please comment in the box below or contact us at admin@ or follow us on Facebook & LinkedIn.
Certified Functional Safety Professional (FSP, TÜV SÜD), Certified HAZOP & PHA Leader, LOPA Practitioner, and Specialist in SIL Verification & Functional Safety Lifecycle, with 18 years of professional experience in Plant Operations and Process Safety across Petroleum Refining and Fertilizer Complexes.
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain






