Octane number is the key parameter to measure the quality and performance of the gasoline fuel. It is the primary specification measurement of gasoline, used worldwide by manufacturers, refiners, and marketers. Current Euro 5, specifications of gasoline fuel require up to 98 RON and 88 MON. Further details about gasoline specifications can be found in the blog “Euro Specifications of Gasoline Fuel”.
Octane rating is the measure of the gasoline fuel’s resistance to knock during combustion in an engine. The higher the resistance higher will be the Octane number. Higher octane number or high resistance to knock will cause efficient combustion of fuel and as a result low knocking by the engine.
Three terms for octane number are used worldwide. The first is the research octane number (RON) and the second is the motor octane number (MON). There is also another term common in the USA, called Anti-Knock Index (AKI) which is an average of RON and MON (R+M/2) but RON is the most common term used.
Advantages of high octane fuel
- Boosts engine performance due to improved fuel compression, as a result, the power of the engine is enhanced for higher efficiency.
- Reduces emissions hence lower pollution to the environment.
- Help to reduce carbon build-up in the engine. Too much carbon build-up in the engine could lead to poor performance.
- Less knocking prevents the chances of damaging the engine.
What is an Engine Knocking?
Low octane fuel, causes knocking and limits the engine to produce the required power. Knocking is caused by the premature ignition of an air and fuel mixture before the spark ignition. This happens when the un-burned air-fuel mixture is compressed in the cylinder and the resulting temperature reaches the auto-ignition point before the ignition by the spark.
Another cause of knocking is the spontaneous explosion of the residual fuel and the air mixture in the combustion chamber as the normal mechanism of combustion nears its end.
Knocking reduces the power of the engine, increases engine wear, and in severe cases may damage the engine.
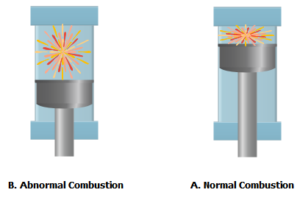
Octane number measurement
The word octane is derived from iso-octane. Octane number is the percentage of iso-octane (2,2,4-trimethylpentane) in the blend of iso-Octane (Octane number 100) and normal-heptane (Octane number 0). For example, a mixture of 90% iso-octane and 10% n-heptane has octane number 90.

ASTM D2699 and ASTM D2700 are standard methods applied to measure the research octane number (RON) under mild conditions of operation at 600 rpm and motor octane number (MON) under severe conditions of operation at 900 rpm respectively.
The Octane number of gasoline is determined in the laboratory tests using a standard single-cylinder test engine having a very sensitive knock meter. The tested fuel knocking is compared with mixtures of iso-octane and n-heptane of various percentages used as a reference. That means if the fuel has the same knocking characteristics as 90 % iso-octane reference fuel then the tested fuel has the octane number of 90.

In an oil refinery, continuous monitoring of RON is required so online analyzers of various types are being used and their working is verified by lab testing.
How to Improve the Octane Number?
- Gasoline is a blend of various gasoline-producing process units of an oil refinery. An increase in the share of high-octane products will improve the RON of the gasoline pool. The following table shows the blend of different naphtha producing plants in an oil refinery.

- Gasoline containing a high proportion of straight-chain alkanes has a low octane rating. However, branched-chain alkanes, cycloalkanes, and aromatic hydrocarbons are much more resistant to knocking. Straight-chain alkanes are converted into isoalkanes in the isomerization unit in the refinery.
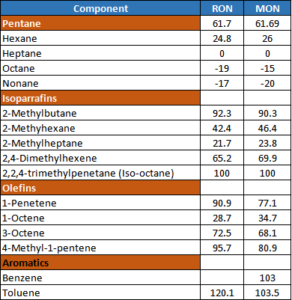
- It can be seen that aromatics or Benzene have an octane number of more than 100. But their concentration is strictly controlled to less than 1% because of its adverse health effects. Benzene is a human carcinogen. It is also present in vehicle exhaust.
- The use of additives like MMT (Methylcyclopentadienyl Manganese Tricarbonyl), methyl tertiary-butyl ether (MTBE), ferrocene, aromatic alcohols, ethanol, and methanol also increase the RON of gasoline. The advantage of adding oxygenates, such as MTBE, methanol, and ethanol, to gasoline is that they cause very little pollution when they burn and are cleaner fuels.
Note: Before the use of Octane additives keeps in mind their impacts on the environment and the vehicle’s engine performance.
For further information, discussion and queries please comment in the box below or contact us at admin@ or follow us on Facebook & LinkedIn.
Certified Functional Safety Professional (FSP, TÜV SÜD Germany), Certified HAZOP & PHA Facilitator, LOPA Practitioner, QRA/FERA and Specialist in SIL Verification & Functional Safety Lifecycle, with 18 years of professional experience in Plant Process Safety across Petroleum Refining and Fertilizer Complexes.
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain







11 thoughts on “Octane Number of Gasoline Fuel”
Good knowledge realted to octane number and its importance to make environment safe……..
Well explained knocking concept