A slurry phase reactor (SPR) is used in petroleum refineries for the hydroprocessing of feeds with very high metal content to obtain lower-boiling products. This technology adopts severe operating conditions and can achieve nearly complete conversion of the residuum while producing finished salable products. Slurry bed reactor-based technologies combine the advantages of carbon rejection technologies in terms of flexibility along with the high-performance achievement due to Hydrogenation. This technology is based on the Bergius Pier technology for coal liquefaction commercialized in Germany since the mid-1930s.
Slurry-phase hydrocracking converts more than 90% residue in the presence of hydrogen under severe process conditions of temperature 450~490°C and pressure 2000~3000 psig. To prevent excessive coking, finely powdered additives made from carbon or iron salts are added to the liquid feed. Inside the reactor, the liquid and powder mixture behaves as a single phase due to the small size of the additive particles.
Heavy refinery streams like residues from deasphalting units, slurry oils from FCC plants, vacuum residues, visbreaking bottom from Visbreaker plants, etc. feedstocks can be converted up to 95% into distillates by applying the slurry phase technology. At 95% conversion, a C5+ yield of approximately 85 wt% is achieved. The C1-C4 yield amounts to 10 wt% with a propane-butane portion of 4 wt%.
Slurry phase hydrogenation is strongly dependent on feedstock quality. Paraffinic crudes such as Amna or Nigerian Medium show the highest hydrogen content and the lowest hydrogen consumption. With the transition to naphthenic and heavy crudes, the hydrogen consumption increases with decreased hydrogen content of the feedstock
Process Description of Slurry Phase Hydrocracking
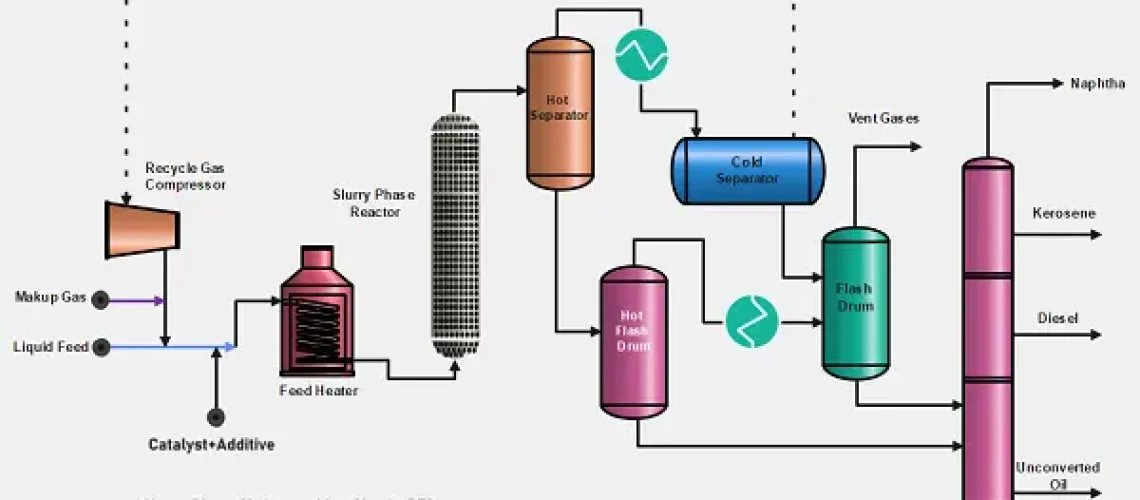
The catalyst and additive (0.1~3.0 wt %) are mixed with the liquid feedstock and then join the hydrogen feed gas. The combined feed and catalyst are preheated with the reactor effluent and finally heated in the fired heater. After that, the feed streams move upward through an empty reactor vessel.
At temperatures in the range of 450~490°C and pressures in the range of 2000~3000 psi, long chain molecules are thermally cracked and this is followed by a reaction of the reaction with hydrogen. The spent catalyst leaves the SPR in a mixture with the liquid product stream and eventually concentrates in the unconverted fraction of the oil.
After leaving the slurry phase reactor the reactor effluent is separated from gases in the hot separator. The gases after cooling are separated from the entrained liquid in a cold separator and then re-compressed and introduced again as a feed gas along with fresh makeup gas.
The Hot and Cold separators liquids are flashed in drums, gases are separated and liquids are sent to the fractionator section. In the fractionator, naphtha, kerosene, and gas oil are separated from the un-converted residue. For further treatment, the separated products are treated in fixed-bed hydrotreaters.
The unconverted nonvolatile fraction at the bottom of the fractionation column, containing all of the catalysts, is recycled back to the reactor, and only a small part of the viscous fraction is purged (1%–3% w/w of the fresh feedstock) to avoid the accumulation of coke precursors and of Ni and V sulfides from the organometallic compounds contained in the feedstock.
With the purge, an equivalent amount of catalyst is fed continuously to the reactor to maintain concentration constant. The purge can be used as a fuel in the cement or steel industries, can be blended with other streams to be used as fuel, and can also be treated in a centrifugal decanter to recover the liquid fraction.

Slurry Phase Reactor
The SPR or SBR (Slurry bed Reactor) is free of internal equipment and operates in a three-phase mode. The solid additive particles are suspended in the primary liquid hydrocarbon phase, through which the hydrogen and product gases flow rapidly in bubble form. Since the oil and catalyst flow concurrently, the mixture approaches a plug flow behavior.
One to three reactors can be employed depending on the feed quality. At the outlet of the reactor, more than 90 % of the feedstock is converted into distillates. At the same time, a major part of the residual oil’s sulfur, nitrogen, and oxygen are converted into hydrogen sulfide, ammonia, and water respectively. Part of the hydrogenation enthalpy is used to preheat the reactor inlet flow to operating temperature, while another part is quenched by cold recycle gas injected through quench nozzles located at the reactor side.
Slurry Phase Reactor Additive
The liquid-phase dispersion inside these reactors is very high. What is important for a stable high conversion operation of the slurry phase reactor shown is the use of an appropriate additive such as carbonaceous material, e.g., lignite coke. A small amount of finely divided powder is used (typically from 0.1 to 3.0 wt%), which can be an additive or a catalyst (or catalyst precursor).
The additive fulfills two major functions. On the one hand, it serves as an adsorbent for metals and destabilized asphaltenes released from the feed material. It thereby performs a carrier function to homogeneously distribute these compounds within the liquid phase, enabling further reaction and transport out of the reactor. To perform this function the additive should show a high specific surface.
Advantages of Slurry Phase Process
- Slurry-phase hydroconversion can be advantageous for the upgrading of heaviest feedstocks due to the remarkably high levels of conversion (>90 vol%) achieved as well as the low costs associated with the catalyst stock and the simple design of the reactor vessel.
- The slurry phase is characterized by improved mass transfer and is thermally more stable.
- SPRs use disposable catalysts with low cost. These slurry catalysts possess high surface area and micron-scale particle size, which means high catalyst utilization.
- The catalyst or additive used in SPR has a larger number of external pores on the surface than the extruded catalyst used in the Fixed bed reactor (FBR), Ebulated bed reactor (EBR), and movable bed reactor (MBR). This difference makes a slurry catalyst less likely to have metal contaminants and coke plugging up these pores, because of their size.
- Spent hydroprocessing catalysts from SPRs are not hazardous wastes and do not require special handling procedures such as those recovered from processes using FBR, EBR, and MBR.
- The smaller size of catalysts used in SPR compared with catalysts employed in other reactors results in smaller distances between particles and less time for a reactant molecule to find an active catalyst site.
- The effectiveness of the dual-role additive (hydrogenation and suppression of coke formation) used in SPRs allows the use of operating temperatures that give high conversion in a single-stage reactor.
- Residue conversion higher than 90% (sometimes higher than 95% and very close to 100%) can be obtained at space velocities much higher than those used for FBR, MBR, or EBR.
- Because of its particular features (empty reactor without internal equipment), the volume of the SPR is maximized.
- The homogeneous phase operation of SPR provides a thermally stable operation (uniform temperature) with no possibility of temperature runaway
Disadvantages of Slurry Bed Reactor
- The main drawback is the extremely poor quality of the unconverted fraction with very high contents of sulfur and metals.
- The poor quality of the unconverted pitch (for example, high metals and sulfur contents) makes it almost impossible to be used as a fuel unless it is blended with other streams.
- Catalyst particle size must be strictly designed to achieve excellent dispersion conditions in order to obtain a high level of conversion.
- SPR has to be carefully designed to maintain a mixed three-phase slurry of heavy oil, fine power catalyst, and hydrogen, and to promote effective contact.
- The effectiveness of the hydroprocessing SPR is highly dependent upon catalyst selection since a slurry catalyst with poor activity will result in coke formation and a biphasic, incompatible product. That means that before commercial use a slurry catalyst has to be subject to different coke suppression and product compatibility tests, among others. Problems of product stability can be prevented by the use of more active catalysts in higher concentrations.
- The inexpensive additives, for instance, Fe-based compounds, or carbonaceous material to control coke formation, show very low activity toward hydrogenation reactions compared with transition metals (molybdenum, nickel, and so on). These low-cost materials can be used in a once-through mode. However, very high hydrogen pressures are necessary to compensate for the scarce catalytic performance. These restrictions have limited the commercialization of hydroprocessing SPR due to the high investment, operating costs, and severity of the process.
- SPR-based hydroprocessing technologies are still on a demonstration scale, and the system has to be demonstrated at a larger scale for refiners to consider this a commercially competitive alternative.
Top References
- Catalytic Hydroprocessing of Petroleum and Distillates by Oballa, Michael C.
- Hydroprocessing of Heavy Oils and Residua by Jorge Ancheyta, James G. Speight
- James G.Speight, in Heavy Oil Recovery and Upgrading
- Deactivation of Heavy Oil Hydroprocessing Catalysts by JORGE ANCHEYTA
Certified Functional Safety Professional (FSP, TÜV SÜD Germany), Certified HAZOP & PHA Facilitator, LOPA Practitioner, QRA/FERA and Specialist in SIL Verification & Functional Safety Lifecycle, with 18 years of professional experience in Plant Process Safety across Petroleum Refining and Fertilizer Complexes.
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain