The crude oil desalting process extracts salts from a crude oil hydrocarbon stream by mixing the stream with a small amount of freshwater forming a water-in-oil emulsion. The resulting emulsion is subjected to an electric field in a crude oil desalter where the water coalesces as underflow from the upper flow of a relatively water-free, continuous hydrocarbon phase. The desalted hydrocarbon stream is produced at a relatively low cost and has a very small residual salt content. The Desalting process removes salts contaminants and suspended solids (soil, Iron oxide, and iron sulfide, clay, sand, etc.) from crude oil before introduction to the crude distillation column.
Crude oil feedstock contains various types of salts, clay, and sand that can not be removed by gravity settling in the storage tanks. Most of the time the salts in the crude oil are in the form of dissolved salts as fine water droplets emulsified in the crude oil. This is called a water-in-oil emulsion, where hydrocarbon is the continuous phase while the dispersed phase is the water. These are mostly magnesium, calcium, and sodium chlorides salts with sodium chloride being the abundant type present in crude oil. These chlorides, except for NaCl, hydrolyze at high temperatures to hydrogen chloride. The Hydrogen chloride produced, dissolves in the overhead system water, producing hydrochloric acid, an extremely corrosive acid.
CaCl2+2H2O= Ca (OH)2+2HCl
MgCl2+2H2O=Mg (OH)2+2HCl
Salts sediments and water are types of Olephobic impurities that can not be dissolved in an oil stream and are removed by applying an electrical desalting process. These impurities are analyzed by BS&W (Base sediments and water) analysis and are measured in PTB pounds per thousand barrels. The typical range of salts, water, and sediments are 5~1000 PTB, 1~2 volume %, 10~500 PTB respectively. The controlled value of Base sediments and water is less than 1 PTB.
Why is desalting of crude oil necessary?
The presence of these salts and sediments poses adverse effects on the refining process. Effective desalting helps to get rid of these damaging effects of salts. The purpose of the desalter is to remove the salts and save the major refinery equipment.
- Salts in the presence of water are responsible for corrosion in the processing system. For example, chloride salts of magnesium generate hydrochloric acid at a temperature above 150°C and may cause severe corrosion both in the liquid and vapor phases.
- Salts deposit inside the furnaces tubes and on the tube bundles of heat exchangers creating fouling, thus reducing the heat transfer efficiency.
- The salts carried with the products act as catalyst poisons in the downstream catalytic hydroprocessing units.
- The presence of inorganic salts in the crude feed will concentrate in the distillation products, thus contaminating the residual petroleum products.
Crude oil desalting Process
In the desalting process of crude oil, preheated crude oil containing salts and water is mixed with fresh water from 3 to 10 volume % water at temperatures from 110 to 150°C. The desired mixing is achieved by using the mixing valve. The salts are dissolved in the wash water and the oil and water phases are separated in a settling vessel by developing a high-potential electrical field. Drops of water are ionized or polarized (one negative side and the other positive) by electric charge and coalesce due to the attraction of opposite charges to form bigger drops of water, which then fall by gravity toward the bottom of the vessel and then drained to the water treatment section. Thus, the emulsion is broken as long as the crude water mixture is in contact with the electrodes. De-emulsifying agents are also injected in small amounts to break the unbroken emulsion.
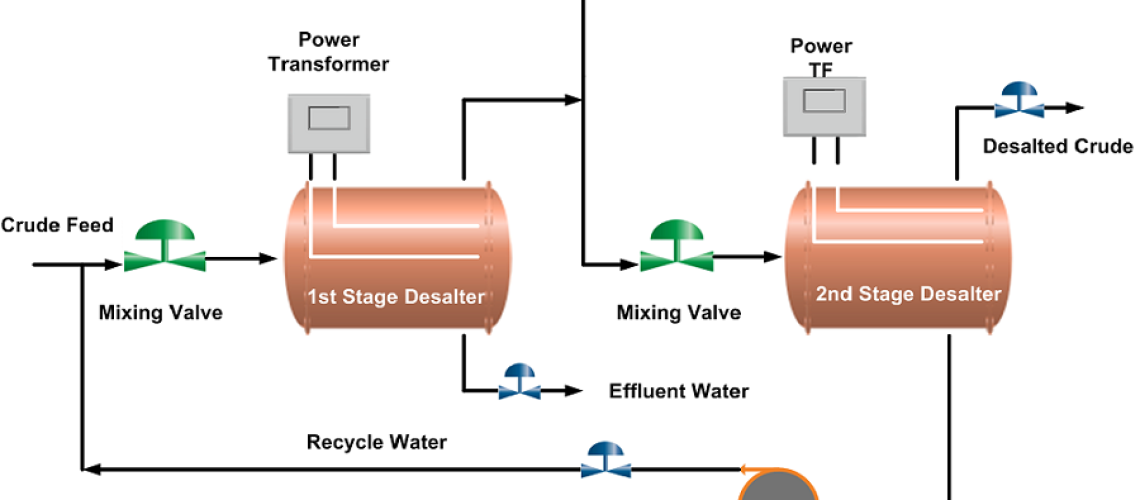
In crude oil desalter, AC or DC current fields may be utilized and potentials from 12,000 to 35,000 volts are used to promote coalescence. Mostly, AC field desalters are used in the refineries. For single-stage desalting up to 90 to 95% efficiencies are obtained while the two-stage desalting process achieves more than 99% efficiency. One process uses a dual field process that uses both the AC and DC fields to provide high dewatering efficiency i.e. up to 99 % in a single stage dual field desalter. The desalters using an AC field are applied near the oil-water interface and a DC field in the oil phase above the interface. About 90% of desalters use AC field separation only. Fig 01 shows the general configuration of the electrical desalter.

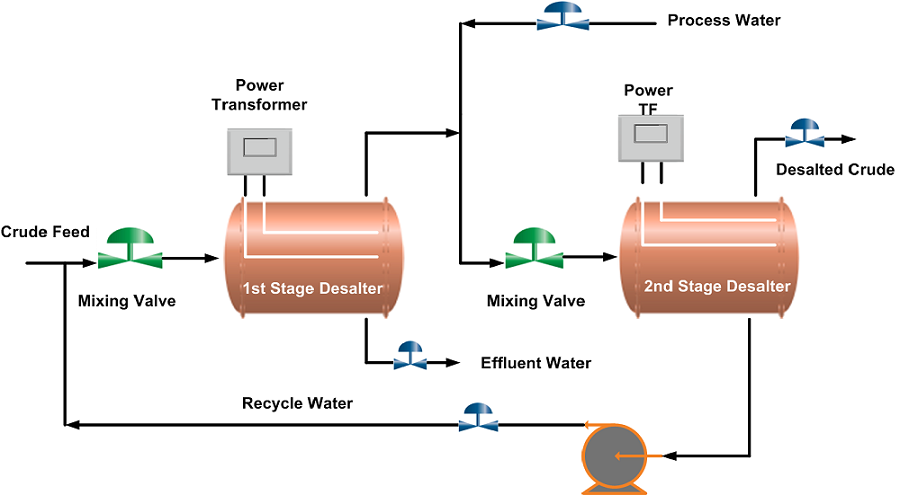
The water level in the desalter is controlled such that the water level should not reach the electrodes, which may cause a short circuit between the electrodes as water is a good conductor of electricity. An interface level transmitter senses the water-oil interface level and sends an alarm signal if the level is high or low and it automatically switches off the power supply in case the water rises above a high level. A low level of water may cause poor desalting and may allow carryover of crude oil with discharged brine to drain. The two-stage desalting process can be seen in the desalting Process Flow Diagram below.
Desalter Operating Variables
For optimized crude oil desalter operation and troubleshooting the desalting process, the following parameters are controlled.
a. Temperature
The settling rate depends on the density and viscosity of the crude. High temperature lowers the density and viscosity, which will increase the water settling rate based on the crude gravity. Higher temperatures may rise vaporization and conductivity, resulting in desalter amperes spikes. Typical desalting temperatures can vary between 100 and 150 °C.
b. Washing Water Flow
Adding water to the crude oil helps in salt removal. Hence, increasing the wash water rate increases the coalescence rate. Low flow of water reduces desalting efficiency because of low contact with brine droplets. Water rates higher than recommended will cause higher power consumption. Depending on the desalting temperature, a minimum value should be used. The typical flow of washing water ranges from 3–8 vol% of the crude feed rate.
c. Water level
Raising the water level reduces the settling time for the water droplets in the crude oil, thus improving the desalting efficiency. However, if the water level gets too high and reaches the lower electrode, it shorts out the desalter. Since the primary electric field depends on the distance between the lower electrode and the water–crude interface, it is always better to keep the level constant for stable operation.
d. Demulsifier Injection Rate
Demulsifiers are basic copolymers with one end being hydrophilic (water absorber and attaches to the surface of the water droplet), and the other end being hydrophobic (loves the oil and is directed to the oil side). When these compounds are adsorbed on the droplet surface, they stabilize the droplet. The demulsifier is added to the crude after the feed pump or before the mixing valve at levels between 3 and 10 ppm of the crude.
e. Pressure Drop Across the Mixing Valve
Mixing the washing water with crude oil is necessary in order to distribute the water and dissolve any suspended salts crystals. Greater the pressure drop across the mixing valve greater will be the mixing of the streams and better separation of the water. On the other hand, over mixing produces fine droplets which tend to stabilize the emulsion and make water separation more difficult. Therefore, there is a compromise in the selection of the appropriate pressure drop across the mixing valve. For optimized operation, a pressure drop between 0.5 and 1.5 bar is used.
f. Desalter Pressure
The operating pressure of the desalter is maintained to avoid vaporization of the liquid phase during desalting. A typical pressure of 12 bar is necessary to achieve this purpose.
g. pH Control
pH of desalter effluent water is normally maintained near 6.5 because at low pH formation of hydrochloric acid from chloride salts and the presence of organic acids in crude oil may cause severe corrosion in the desalter drum. Better water removal is obtained in electrical desalters when they are operated at a pH of 6. If the pH of the brine exceeds 7, emulsions can be formed because of the sodium naphthenate and sodium sulfide present. The caustic solution is injected with the incoming water to control the pH aqueous phase.
h. Voltage
If the voltages go up and current flow becomes excessive then the desalting operation becomes costly. On the other side, if the voltage drops then demulsification also drops down, resulting in poor desalting efficiency.
For further discussion and comments please comment in the comment box below or contact at admin@theptrosolutions.com
Certified Functional Safety Professional (FSP, TÜV SÜD), Certified HAZOP & PHA Leader, LOPA Practitioner, and Specialist in SIL Verification & Functional Safety Lifecycle, with 18 years of professional experience in Plant Operations and Process Safety across Petroleum Refining and Fertilizer Complexes.
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain
- Nasir Hussain