Crude oil is a naturally occurring unrefined liquid petroleum, a mixture of hydrocarbons (Compounds mainly of Hydrogen and Hydrocarbon) that is found accumulated underground in the earth’s crust. Various types of crude oils with different properties are available in the market. Its price depends upon density, sulfur, metals, and other contaminants. The composition of crude oil varies with the geographical location, age, and depth of the well.
Hydrocarbon (Hydrogen and Carbon) content in crude can be as high as 97% in lighter Paraffinic crude and as low as 50 % in heavy and Bitumen crude. Heavy crude oils are difficult to operate and pose challenges to the refinery operation. Knowing about crude oil is not only necessary to optimize the refinery operation but also for designing new plants. For more about crude oil view the blog “Crude Oil Classification and Benchmars”
The composition of crude oil can be categorized based on the presence of elements, molecules, and compounds present in the crude oil. Further, crude oil was analyzed as mentioned in the Crude Oil Assay, to find the components (like Sulfur, Metals, and Ashpaltics), and petroleum products yield for, oil traders, producers, and designers enabling them to predict the true value of crude oil for a specific facility.
1. Element-Based Composition
Complete elemental analysis is the first step in determining the quality and general behaviour of crude oil. Whatever the source is, with fewer exceptions, the proportions of the elements remain nearly the same. More the composition of C atoms heavier will be the crude. The typical range of these elements is shown in the chart below;

4. Chemical Composition of Crude Oil
Crude oils are characterised by the type of hydrocarbon compounds present in them that is most common molecules are paraffins, naphthenes and aromatics. Paraffins are the most common hydrocarbons in crude oil; certain liquid paraffins are the major element of gasoline and therefore bear high market value. Naphthenes are an important part of all liquid refinery products, but they also form some of the heavy asphaltic-like residues of refinery processes. Aromatics generally are only a small percentage of most crudes. The most common aromatic in crude oil is Benzene, a popular petrochemical industry feed. Their typical ranges are given in the table and described below.
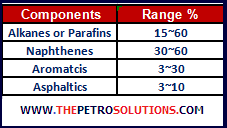
Paraffin or Alkanes
These are the most common hydrocarbons in crude oil and are saturated having the general formula CnH2n+2. They can be straight-chain (n-paraffins), branched-chain (iso-paraffins), or cyclic alkanes. All the Carbon items have a single bond.
Examples are; Methane (CH4), Ethane (C2H6), Propane (C3H8), etc. The first four compounds are in the gas form at atmospheric pressure while C5H10 (Pentane) is in the liquid state followed by Hexane (C6H14), Heptane (C7H16), Octane (C8H18), and so on.

Naphthenes or Cycloalkanes
These are also called Cycloalknes and are saturated Hydrocarbons but have at least one ring of carbon atoms having the general formula CnH2n.
The boiling point and densities of Naphthenes are higher than those of alkanes having the same number of carbon atoms. These are commonly present in crude oil as rings with five or six carbon atoms. These rings usually have alkyl substituents attached to them. Multi-ring naphthenes are present in the heavier parts of the crude oil.

a. Aromatics
These are unsaturated HC with at least one Benzen ring. The simplest aromatic compounds are Benzene and each aromatic has one or more Benze rings. A greater number of Benzene rings make the crude oil heaver and are called Polynuclear aromatic compounds. Higher concentration of Aromaicts, particularly heavy aromatics causes coking and catalyst deactivation in processing.

b. Sulfur
Crude oil with less than 1 % is called Sweet crude and greater than 1 % is called Sour crude. Crude oil contains sulfur heteroatoms in the form of elemental sulfur S, inorganic forms, and most importantly organic forms, in which sulfur atoms are positioned within the organic hydrocarbon molecules. Examples are mercaptans, sulfides, and polycyclic sulfides. In general, heavier crude carries more Sulfur.

c. Nitrogen
Crude oils contain minor amounts of nitrogen compounds. Nitrogen compounds are more stable than sulfur compounds and are difficult to remove. They can be responsible for the poisoning of a hydrocracking catalyst, and they also contribute to gum formation in finished products.

The nitrogen compounds in crude oils may be classified as basic or non-basic. Basic nitrogen compounds consist of pyridines. The greater part of the nitrogen in crude oils is the non-basic nitrogen compounds which are generally of pyrrole types.
d. Oxygen
Oxygen in crude oil can be found in various forms. These include alcohols, ethers, carboxylic acids, phenolic compounds, ketones, esters, and anhydrides. The presence of such compounds causes the crude to be acidic causing corrosion during processing.

e. Metals
Present in minor concentrations but cause significant problems in processing. Major metals include Nickel, Vanadium, Iron, Arsenic, lead, etc. These metallic compounds cause poisoning of the hydroprocessing and hydrocracking catalysts.
Part of the metallic constituents of crude oil exists as inorganic water-soluble salt, mainly as chlorides and sulfates of sodium, potassium, magnesium, and calcium. These are removed in desalting operations.
f. Asphaltics
This is the heaviest component in crude oil. Its presence in high amounts can create massive problems during processing. They may also create transportation problems due to high gravity and viscosity. They cause coke formation and metal deposition on the catalyst surface causing its deactivation.
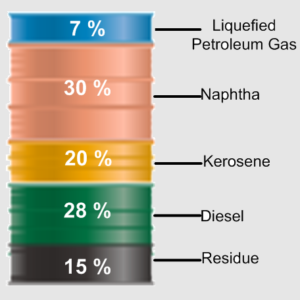
3. Composition based upon Petroleum Products Yield
The crude oil can also be analyzed to find the percentage of petroleum products (Naphtha, Kerosene, Light Diesel, Heavy Diesel, and Bottom) present in the crude oil. This type of composition is critical for the smooth operation of the oil refinery and for designing a new refining facility. An example of this composition can be seen below.
For further information, discussion and queries please comment in the box below or contact us at admin@ or follow us on Facebook & LinkedIn.




13 thoughts on “Composition of Crude Oil”
Good information about crude oil. Also write about curd oil properties and classification.
Please write some thing more detailed about Asphaltics.
Dear,my coming blogs are also about crude oil. I will try to focus on Asphaltics as well.
Very good information about components of crude oil.
Thank you.